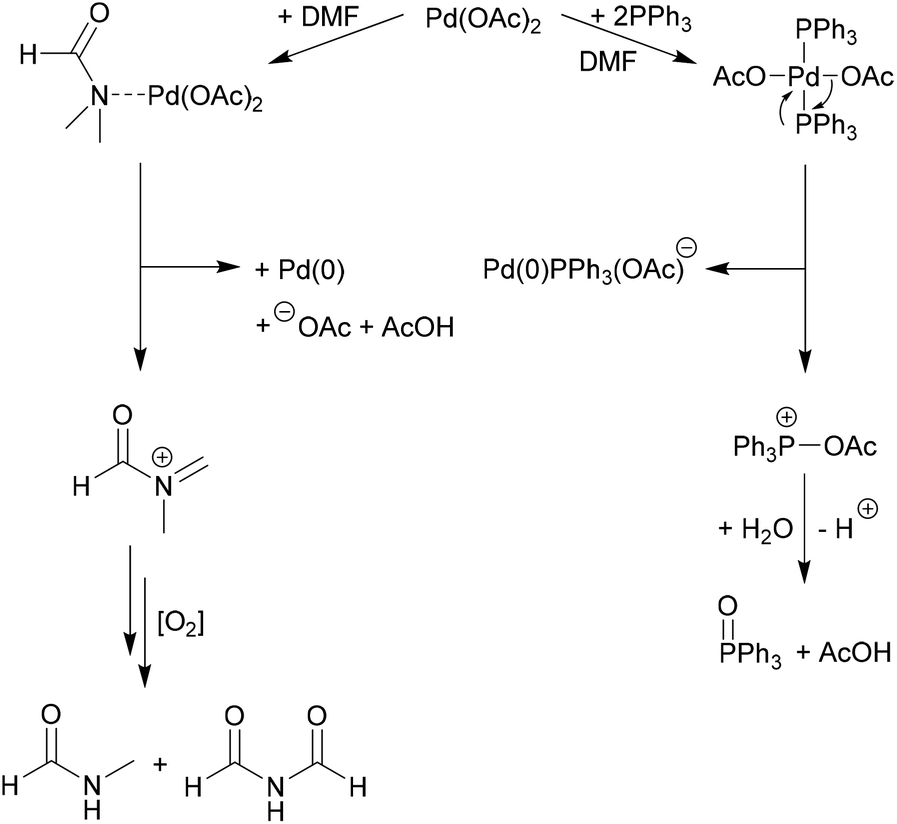
The Palladium Acetate‐Catalyzed Microwave‐Assisted Hirao Reaction without an Added Phosphorus Ligand as a “Green” Protocol: A Quantum Chemical Study on the Mechanism - Keglevich - 2017 - Advanced Synthesis & Catalysis -
Structural characterisation of solution species implicated in the palladium -catalysed Heck reaction by Pd K-edge X-ray absorption spectroscopy : pall ... - Journal of the Chemical Society, Dalton Transactions (RSC Publishing) DOI:10.1039/B200617K
The ubiquitous cross-coupling catalyst system 'Pd(OAc)2'/2PPh3 forms a unique dinuclear PdI complex: an important entry poin

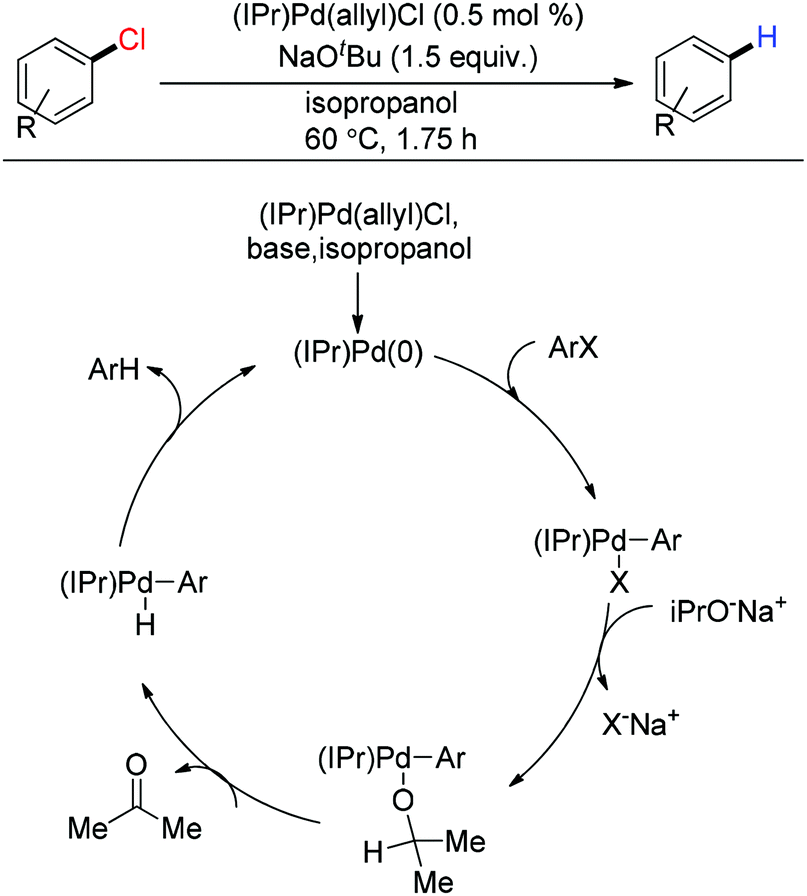
The Impact of Palladium(II) Reduction Pathways on the Structure and Activity of Palladium(0) Catalysts - Wei - 2013 - Angewandte Chemie International Edition - Wiley Online Library

Palladium-catalyzed microwave-assisted Hirao reaction utilizing the excess of the diarylphosphine oxide reagent as the P-ligand; a study on the activity and formation of the “PdP2” catalyst

US20160340311A1 - Recovery and/or reuse of palladium catalyst after a suzuki coupling - Google Patents

The Palladium Acetate‐Catalyzed Microwave‐Assisted Hirao Reaction without an Added Phosphorus Ligand as a “Green” Protocol: A Quantum Chemical Study on the Mechanism - Keglevich - 2017 - Advanced Synthesis & Catalysis -
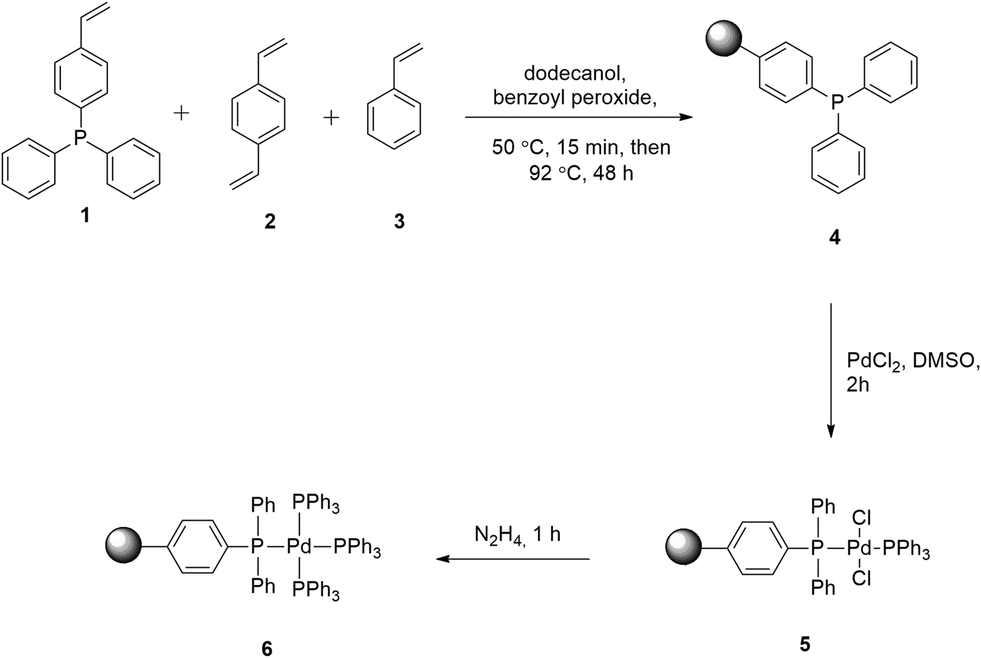
Immobilized tetrakis(triphenylphosphine)palladium(0) for Suzuki–Miyaura coupling reactions under flow conditions - Reaction Chemistry & Engineering (RSC Publishing) DOI:10.1039/C8RE00235E

Polymer Supported Triphenylphosphine-Palladium Acetate Complex PS-TPP-Pd(OAc)2 as a Heterogeneous and Reusable Catalyst for Indirect Reductive Amination of Aldehydes – topic of research paper in Chemical sciences. Download scholarly article PDF and read
![14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation 14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation](https://labchem-wako.fujifilm.com/sc/01/14221-01-3.png)
14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation

US20160340311A1 - Recovery and/or reuse of palladium catalyst after a suzuki coupling - Google Patents

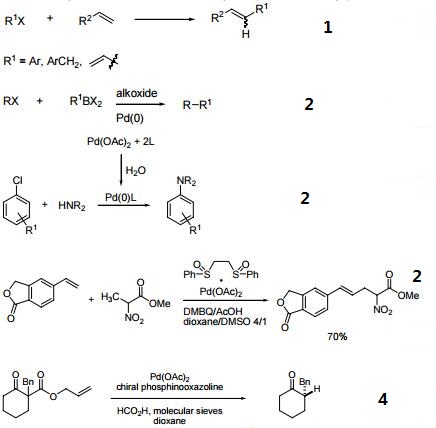
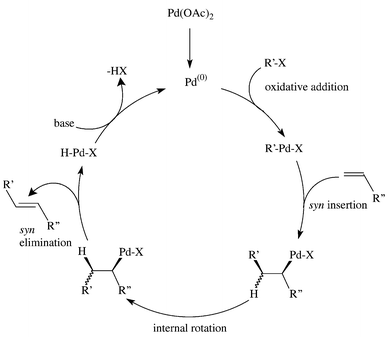
SciELO - Brasil - Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the