![PDF] Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction | Semantic Scholar PDF] Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/fc005877bed22b9a856c6808d6652c3b503c590b/3-Table1-1.png)
PDF] Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction | Semantic Scholar

Selective Incorporation of Fluorine in Pyrazoles - Sloop - 2015 - European Journal of Organic Chemistry - Wiley Online Library
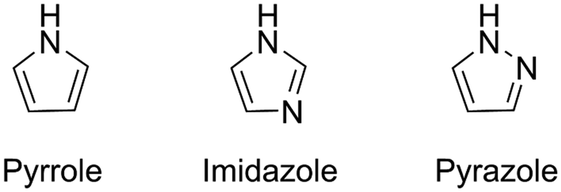
Reactions of pyrrole, imidazole, and pyrazole with ozone: kinetics and mechanisms - Environmental Science: Water Research & Technology (RSC Publishing) DOI:10.1039/C9EW01078E
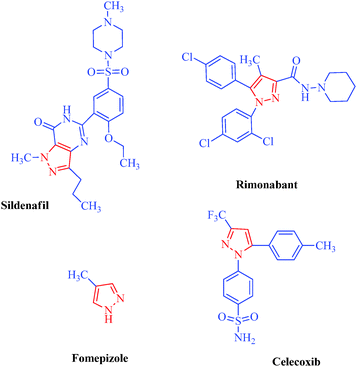
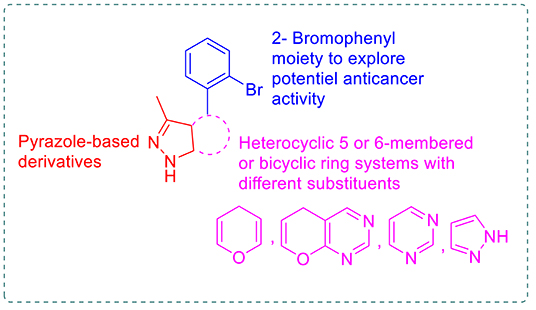
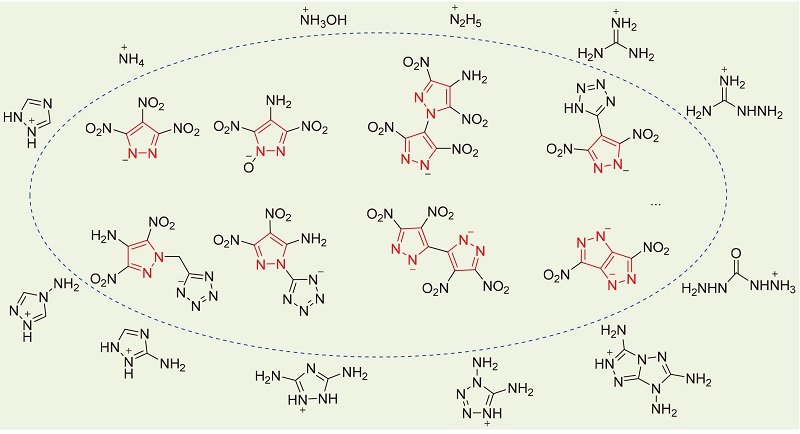
Review: biologically active pyrazole derivatives - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C6NJ03181A
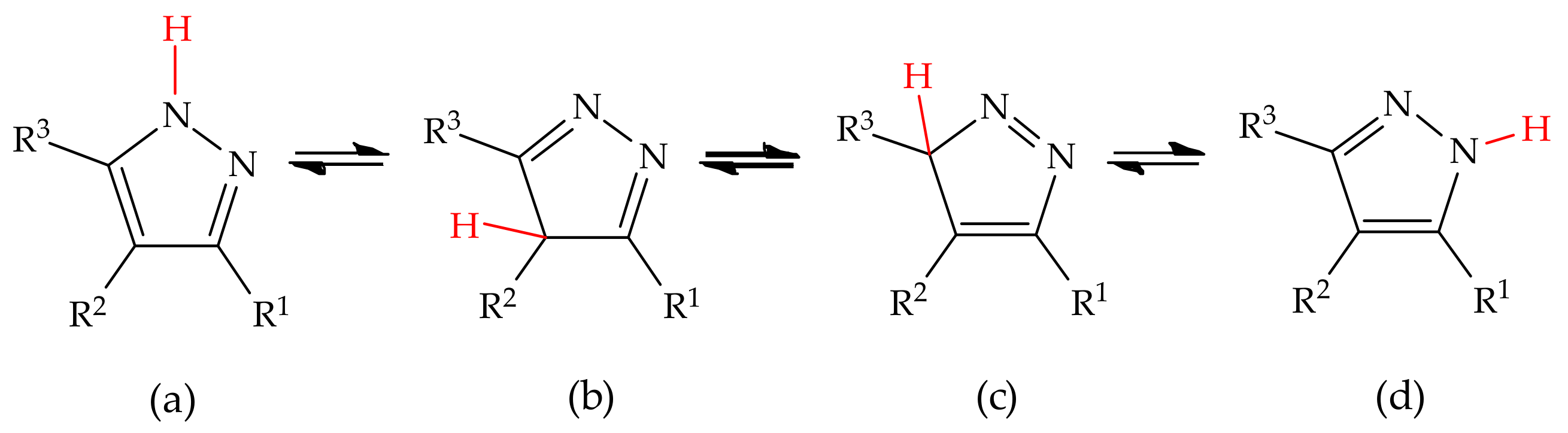
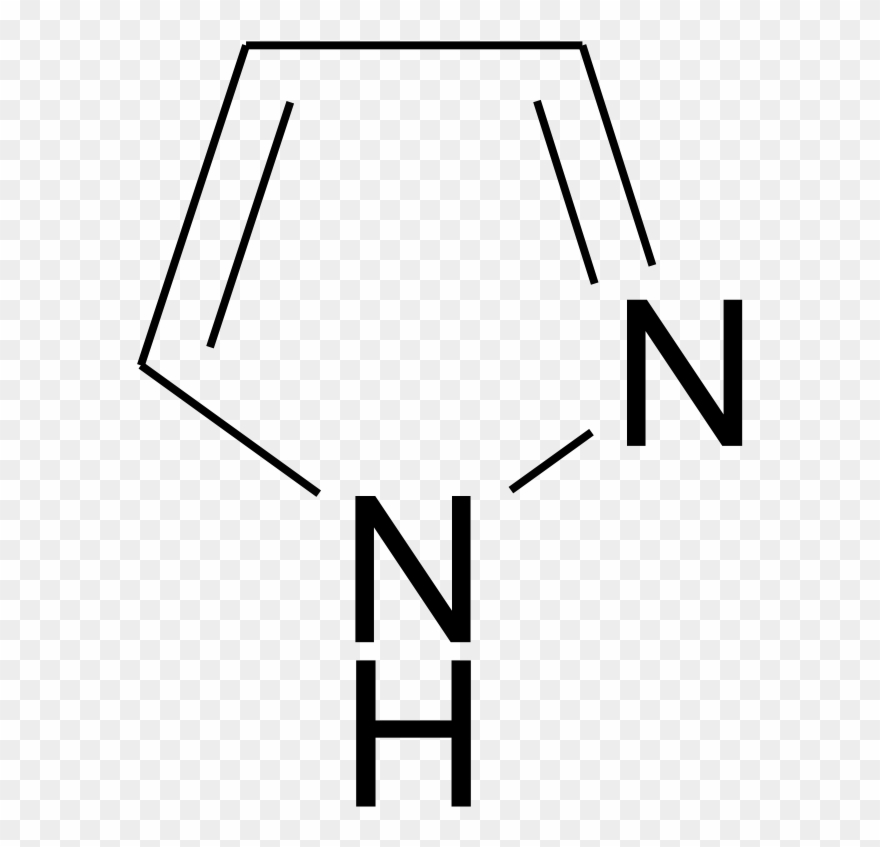
Molecules | Free Full-Text | Revisiting the Structure and Chemistry of 3(5)-Substituted Pyrazoles | HTML
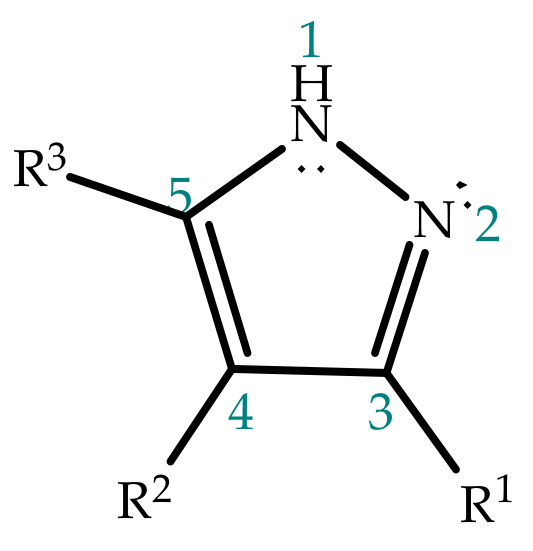
Molecules | Free Full-Text | Revisiting the Structure and Chemistry of 3(5)-Substituted Pyrazoles | HTML
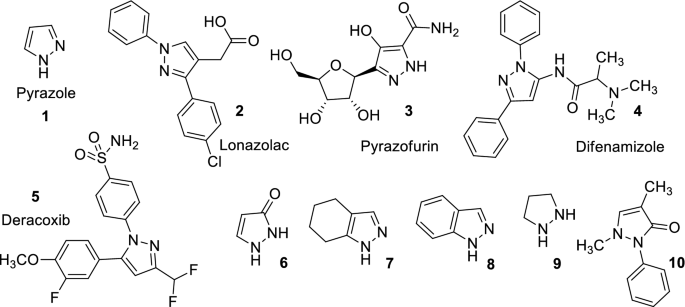
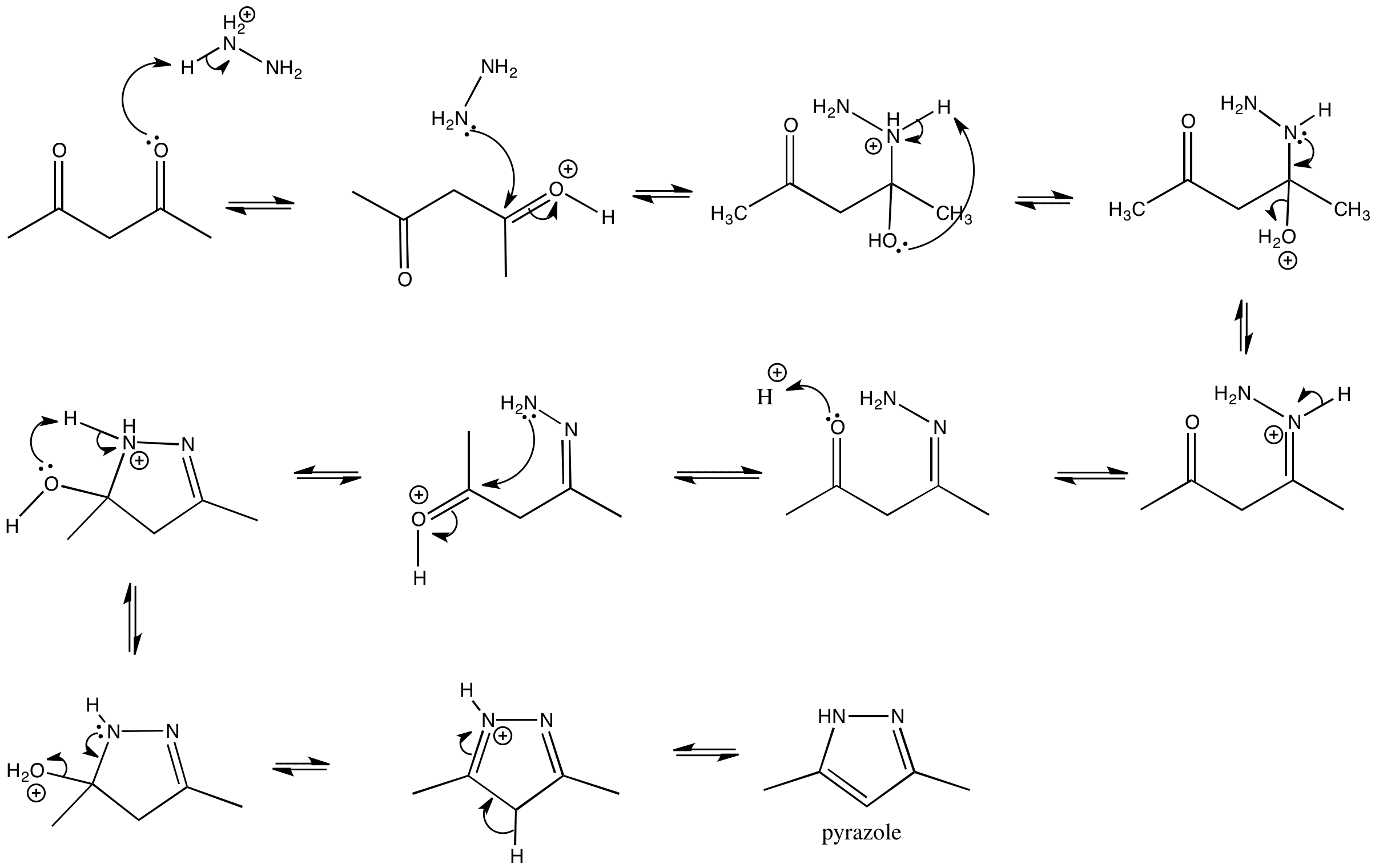
Recent developments in synthetic chemistry and biological activities of pyrazole derivatives | SpringerLink

Facile Access to 3,5‐Dihalogenated Pyrazoles by Sydnone Cycloaddition and their Versatile Functionalization by Pd‐Catalyzed Cross‐Coupling Processes - Delaunay - 2011 - European Journal of Organic Chemistry - Wiley Online Library

Synthesis of Nitriles – Synthesis of 4-Cyano Pyrazole, 5-Aminopyrazole Derivatives and the Deamination of 5-Aminopyrazole Derivatives | IntechOpen