PLOS Medicine: Efficacy and Safety of the RTS,S/AS01 Malaria Vaccine during 18 Months after Vaccination: A Phase 3 Randomized, Controlled Trial in Children and Young Infants at 11 African Sites

Randomized, Double Blind and Placebo Controlled Study Using a Combination of Two Probiotic Lactobacilli to Alleviate Symptoms and Frequency of Common Cold

CONSORT diagram. Composition of the per protocol population with sleep... | Download Scientific Diagram
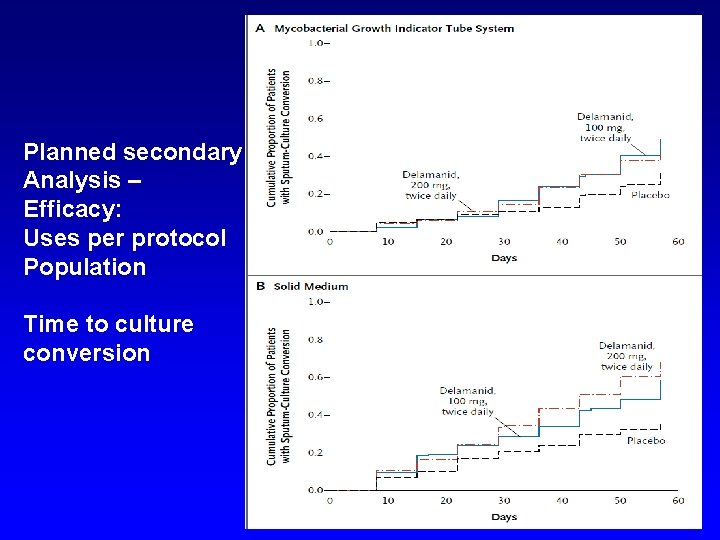
PLOS ONE: An Open-Label, Randomised Study of Dihydroartemisinin-Piperaquine Versus Artesunate-Mefloquine for Falciparum Malaria in Asia

Precise definitions of some terminology for longitudinal clinical trials: subjects, patient populations, analysis sets, intention to treat, and related terms. | Semantic Scholar
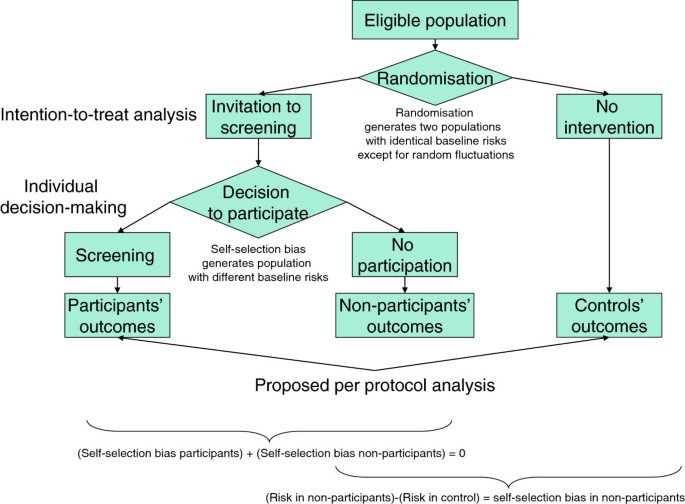
Screening: the information individuals need to support their decision: per protocol analysis is better than intention-to-treat analysis at quantifying potential benefits and harms of screening | BMC Medical Ethics | Full Text

CONSORT Flow Diagram. *Per protocol population was used for applicable... | Download Scientific Diagram

Supplemental Materials for Hepatic arterial infusion of oxaliplatin plus raltitrexed in patients with intermediate and advanced stage hepatocellular carcinoma: A phase II, single-arm, prospective study - European Journal of Cancer

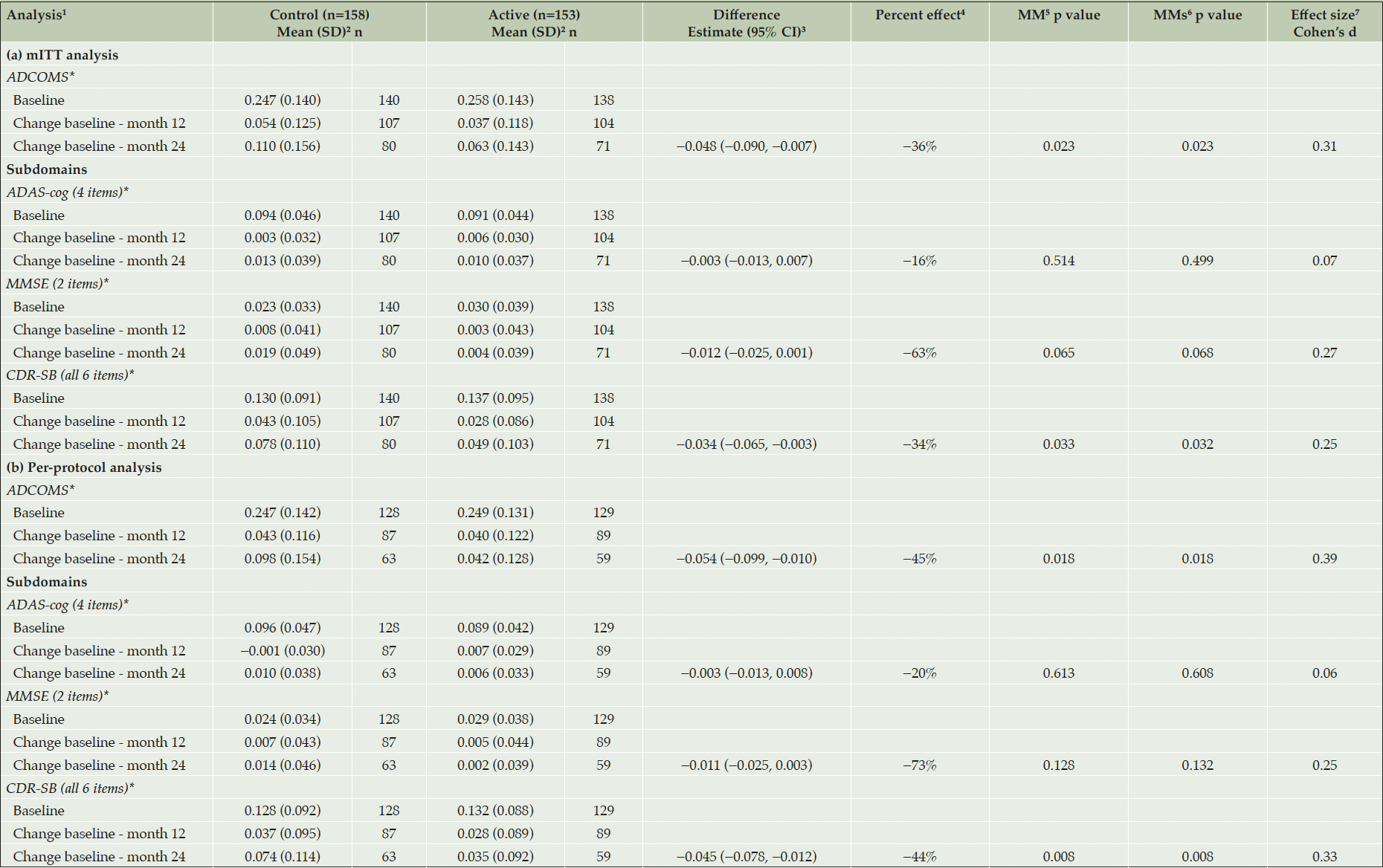
Predicting the Placebo response in OA to Improve the Precision of the Treatment Effect Estimation - Tools4Patient

New regimens of benznidazole monotherapy and in combination with fosravuconazole for treatment of Chagas disease (BENDITA): a phase 2, double-blind, randomised trial - The Lancet Infectious Diseases

A Prospective, Randomized, Open-Label Trial of Early versus Late Favipiravir Therapy in Hospitalized Patients with COVID-19 | Antimicrobial Agents and Chemotherapy