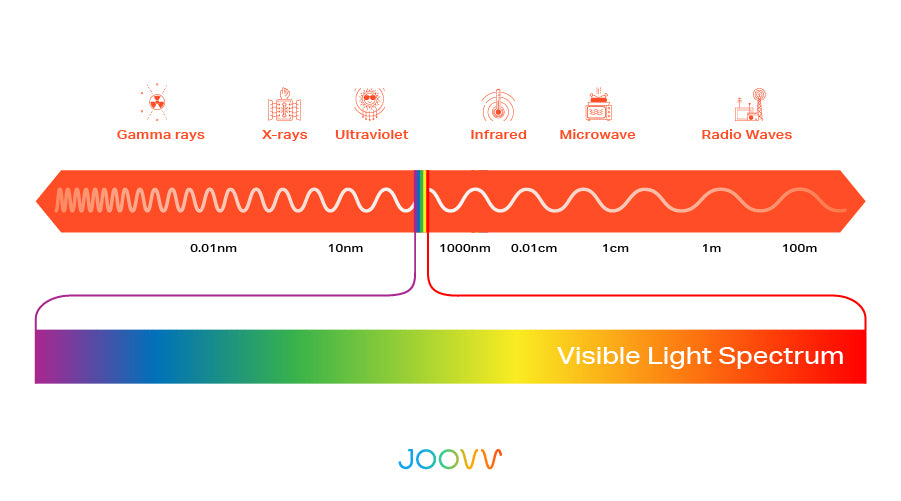
vision - Is the yellow we perceive when our eyes are hit by red and green light at the same time the same yellow that is at the yellow frequency/wavelength? - Physics
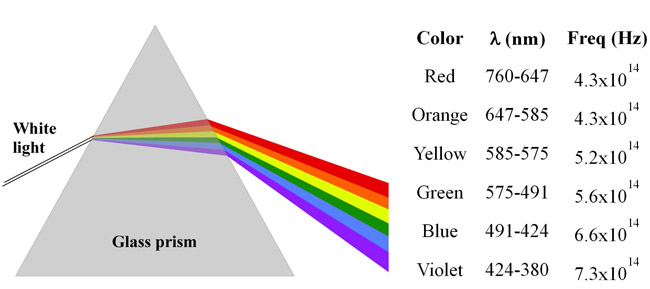
A metal foil has a threshold frequency of 5.45× 1014 Hz. Which of the colors of visible light have enough energy to eject electrons from this metal? | Socratic

WHAT DO WE KNOW ABOUT LIGHT?. What is Light? Light is a wave that we can see. –Light can carry heat and warmth. –Light has color. –Light can be bright. - ppt

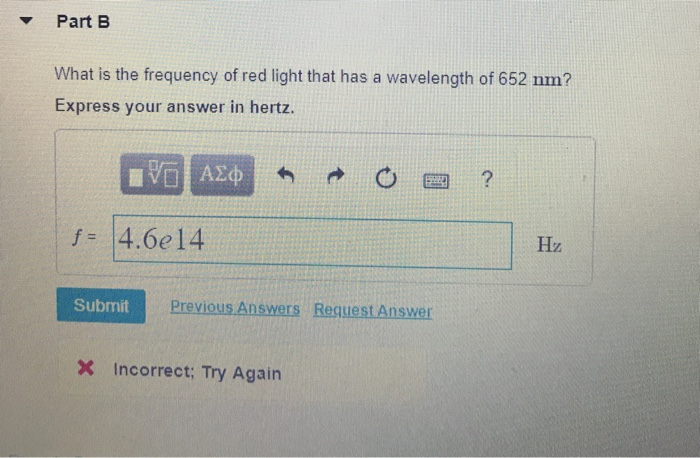
Calculate the frequency of the red light emitted by a neon sign with a wavelength of 690 nm - Brainly.com

Using the knowledge of 'Electromagnetic spectrum', explain why traffic lights contain only Green, Red and Orange. | Homework.Study.com

Do 3 light sources with same intensities, but different wavelengths(blue,green, red) provide same amount of energy for photosynthesis? | ResearchGate

How Fast Would You Have to Approach a Red Traffic Light in Order to See it as Green? (Whiteboard Sketch #2) | scientific finger food

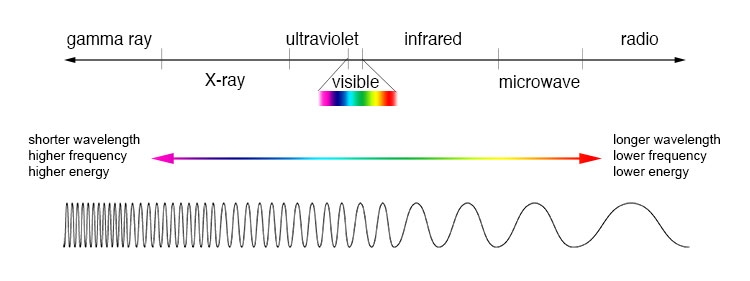
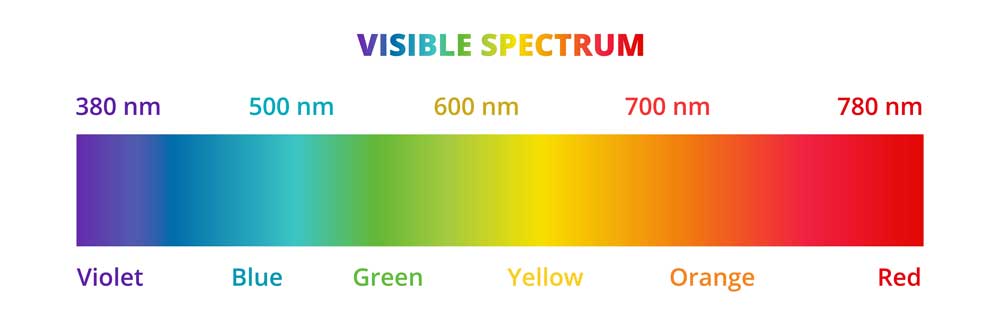
:max_bytes(150000):strip_icc()/the-visible-light-spectrum-2699036_FINAL2-c0b0ee6f82764efdb62a1af9b9525050.png)