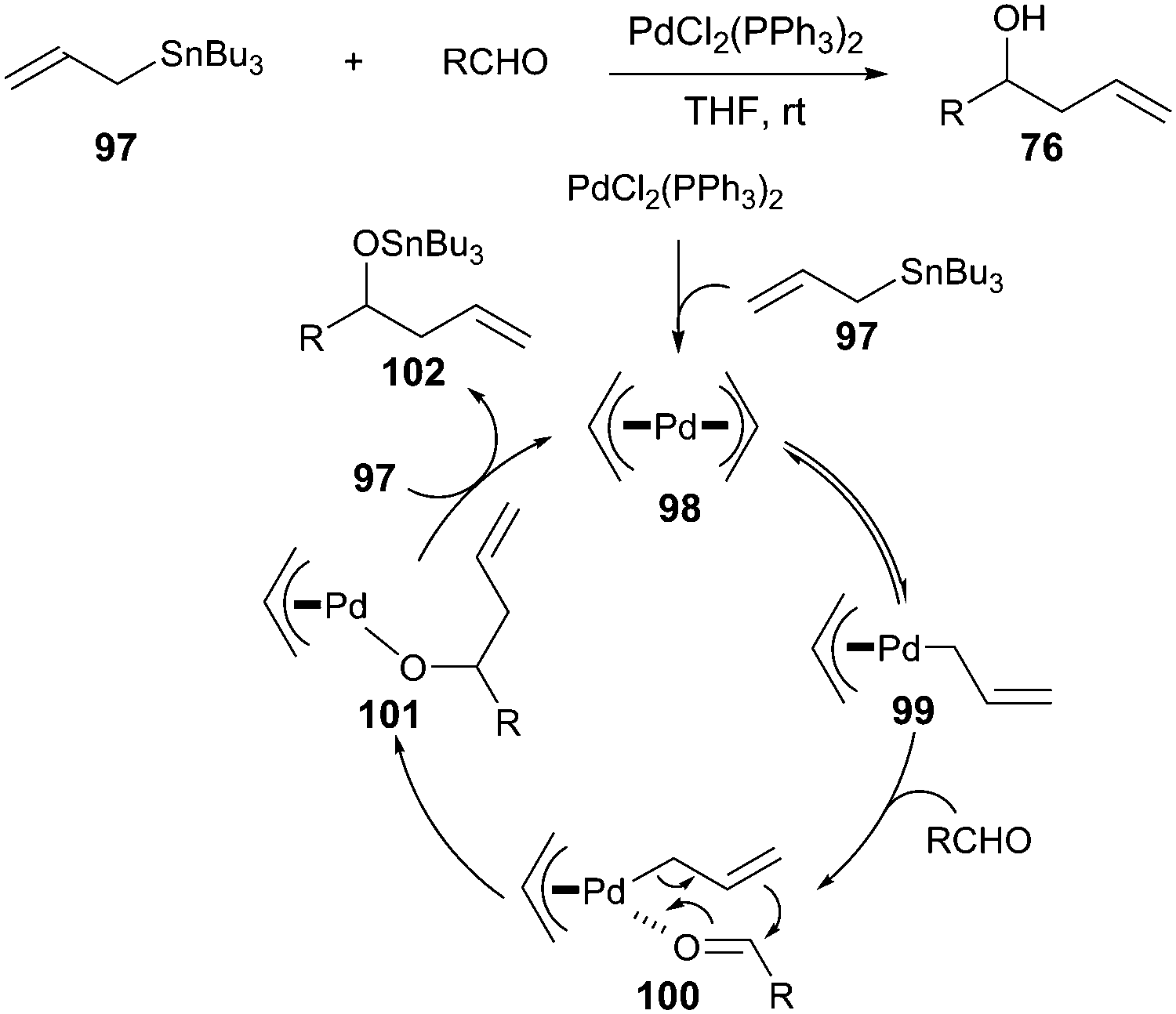
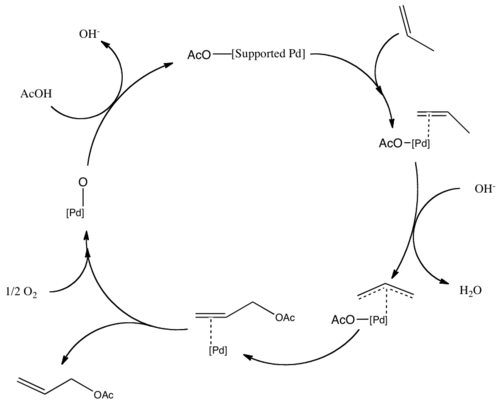
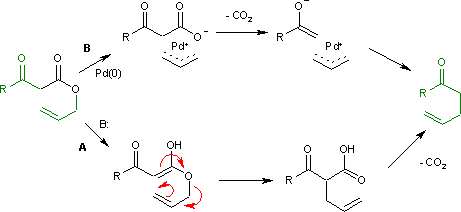
Pd-catalyzed asymmetric allylic alkylations <i>via</i> C-H activation of <i>N</i>-allyl imines with glycinates. - Abstract - Europe PMC

Nickel-catalyzed allylic carbonylative coupling of alkyl zinc reagents with tert -butyl isocyanide | Nature Communications
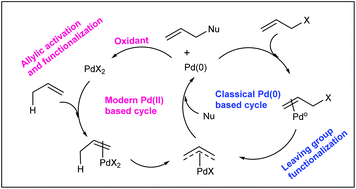
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds
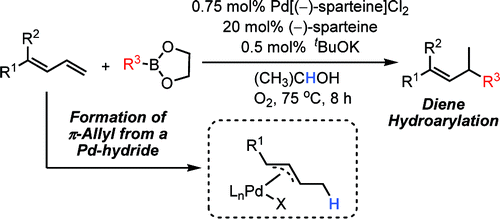
Palladium-Catalyzed Hydroarylation of 1,3-Dienes with Boronic Esters via Reductive Formation of π-Allyl Palladium Intermediates under Oxidative Conditions,Journal of the American Chemical Society - X-MOL

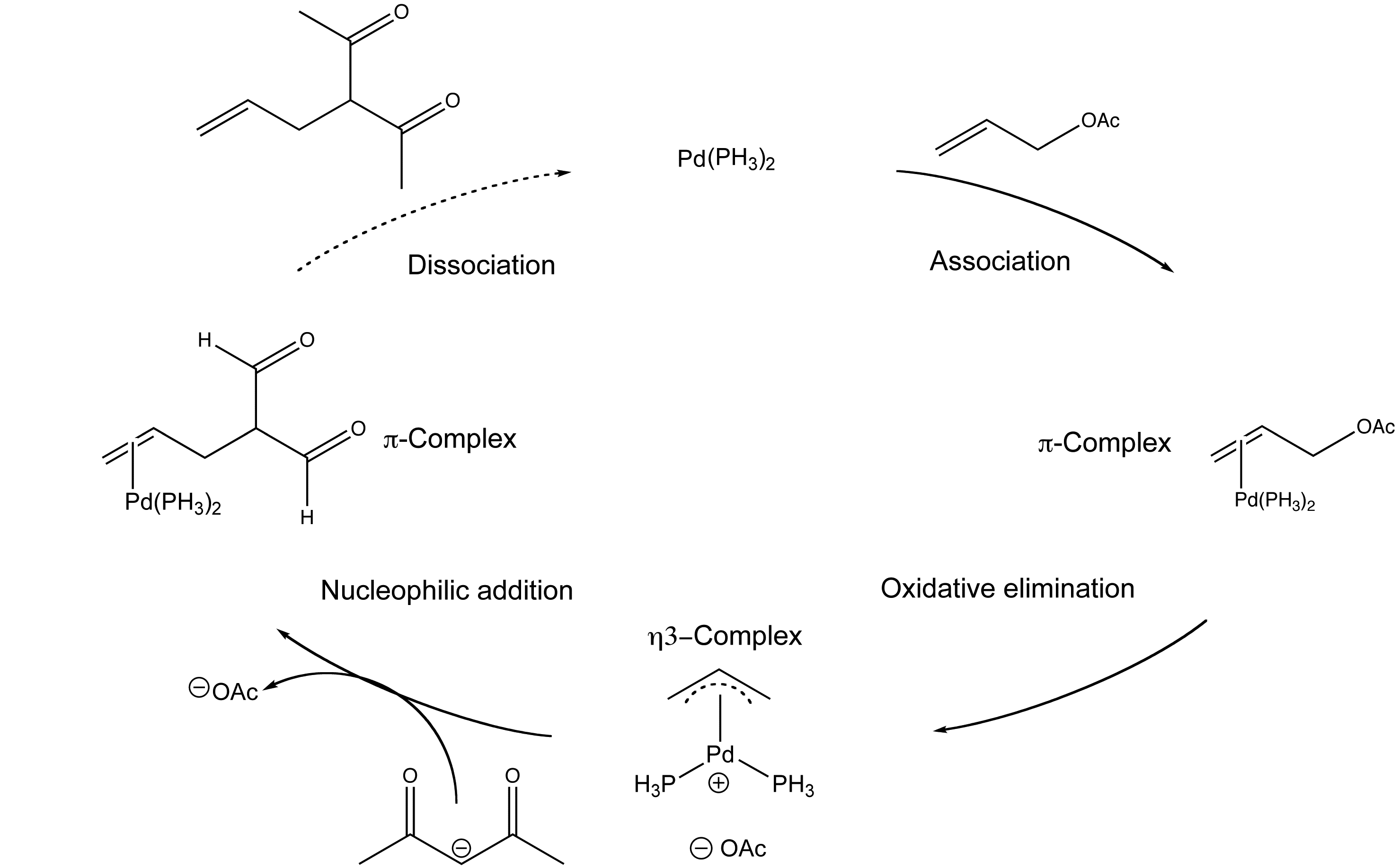
Mechanism of allyl deprotection through catalytic palladium π-allyl... | Download Scientific Diagram

Figure 1 from Amide α,β-Dehydrogenation Using Allyl-Palladium Catalysis and a Hindered Monodentate Anilide. | Semantic Scholar

Palladium-Catalyzed Asymmetric Reduction of Racemic Allylic Esters with Formic Acid: Effects of Phosphine Ligands on Isomerization of π-Allylpalladium Intermediates and Enantioselectivity - ScienceDirect

Irreversible Catalytic Ester Hydrolysis of Allyl Esters to Give Acids and Aldehydes by Homogeneous Ruthenium and Ruthenium/Palladium Dual Catalyst Systems - Nakamura - 2011 - Advanced Synthesis & Catalysis - Wiley Online Library

A Novel, One-Step Palladium and Phenylsilane Activated Amidation from Allyl Ester on Solid Support – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka
A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions
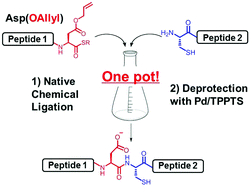
Efficient peptide ligation between allyl-protected Asp and Cys followed by palladium-mediated deprotection - Chemical Communications (RSC Publishing)

CpRuII-chiral bisamidine complex catalyzed asymmetric Carroll-type decarboxylative allylation of β-keto allyl esters - ScienceDirect

Synthesis of 1,3‐Diarylpropenes through Palladium‐Catalyzed Mizoroki–Heck and Allyl Cross‐Coupling Reactions Using Hydrazones as Ligands - Mino - 2012 - European Journal of Organic Chemistry - Wiley Online Library