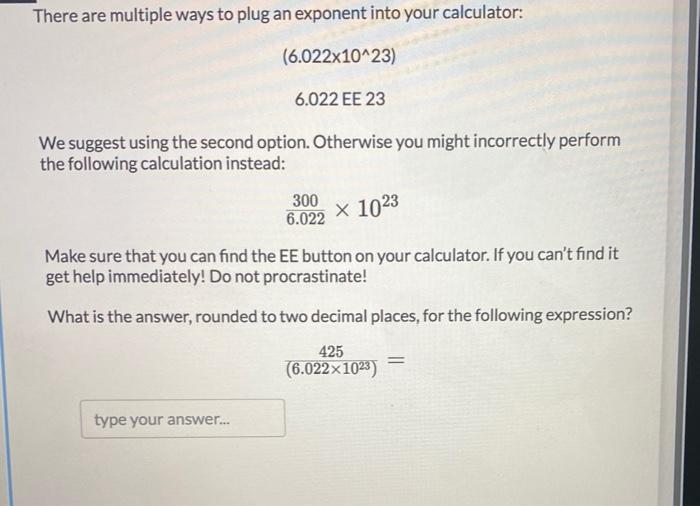
What is the mass in grams of : (i) 6.022 xx 10^(23) atoms of oxygen (ii) 1.0 xx 10^(23) molecules of H(2)S (iii) 6.022 xx 10^(23) molecules of oxygen ?

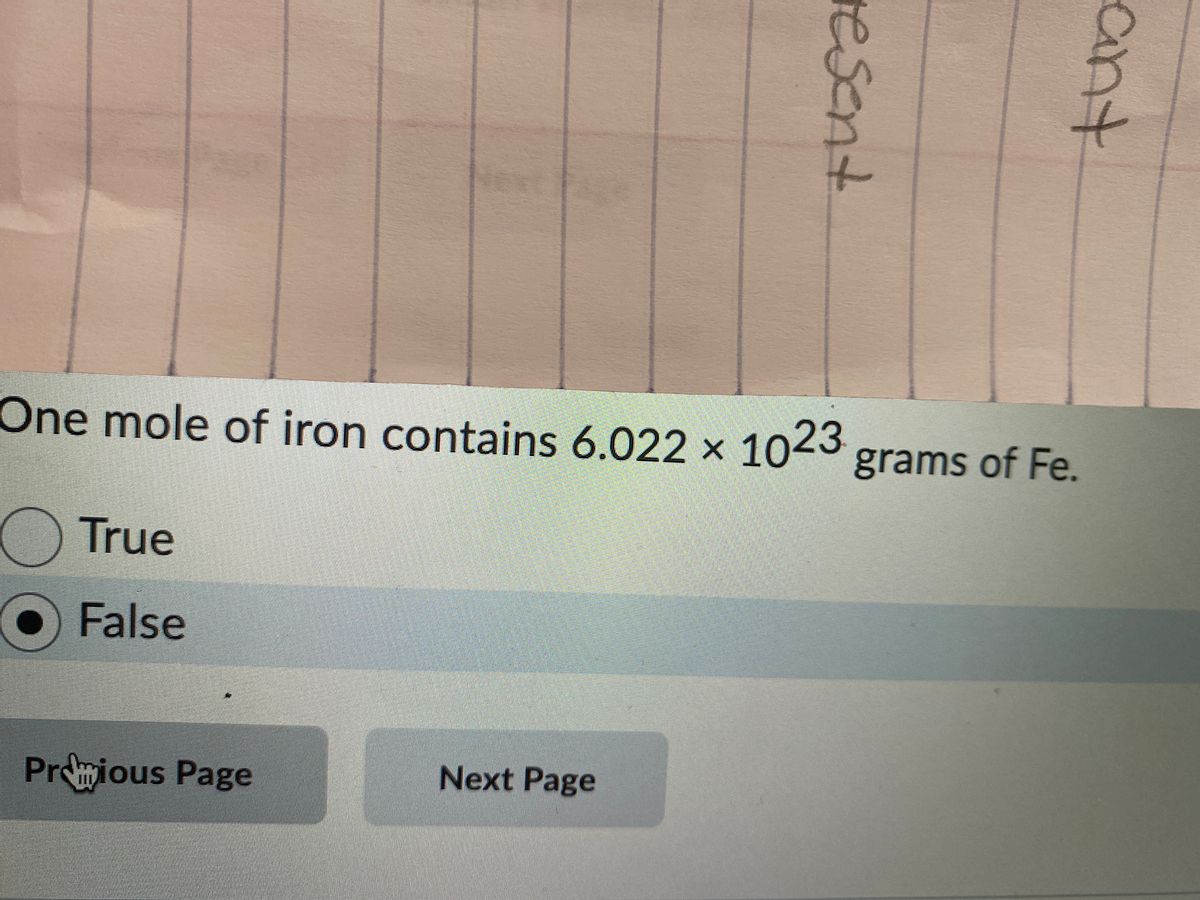
12 g C - 12 contains 6.022 × 10^23 atoms of carbon.(a) 6.022 × 10^23 is known as .............(b) Calculate the number of carbon atoms present in 48 g C - 12.(c)

If Avogadro number NA is changed from 6.022 × 10^23 mol^-1 to 6.022 × 10^20 mol^-1 , this would change :
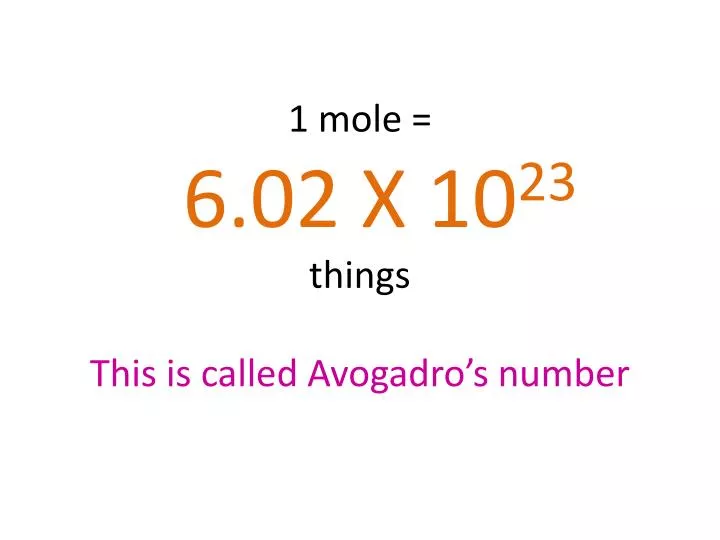
PPT - 1 mole = 6.02 X 10 23 things This is called Avogadro's number PowerPoint Presentation - ID:4272623

Question Video: Determining the Number of Oxygen Atoms Present in a Given Number of Moles of Aluminum Nitrate | Nagwa

Calculate the mass of 6 022 x 10^23 molecules of CaCO3 - Science - Atoms and Molecules - 13283691 | Meritnation.com

SOLVED: Choose the items below that are equivalent to 6.022 x 1023 particles...(particles = atoms, molecules, or formula units) a. 1 mole of everything b. 26.98g of Al c. 58.44g of NaCl

Regal Tutors - 6.022 x 10^23 is Avogadro's number. It tells us how many particles are in one mole of a substance. For example, one mole of carbon has 6.022 x 10^23